PHYS 1020 - Physics of Energy
Unit 2: Thermodynamics
In this second unit, we’ll look at our first and most basic use of energy: heating. To understand this, we need to learn about thermodynamics:
- Thermo- = heat
- -dynamics = motion
After we discuss thermodynamics, we talk about using heat to harness energy. In particular, we will look at the use of fossil fuels.
Thermal Energy
Reading:
- Textbook sections 5A–5C
Materials:
- Adiabatic Compression device
The First Two Laws of Thermodynamics
There are four basic laws of thermodynamics. We’ll learn them over the next few days.
Zeroth Law of Thermodynamics
If two objects are in thermal contact (energy can flow between them), then eventually they will reach thermal equilibrium (equal energy).
The zeroth law of thermodynamics states:
If two objects are each in thermal equilibrium with a third object, then they will be in thermal equilibrium with each other.
A simpler example:
- If Block A is in thermal equilibrium with Block B
- And Block C is also in thermal equilibrium with Block B
- Then Block A is in thermal equilibrium with Block C
This is a very intuitive law and almost does not need to be stated. However, this law is the reason we can use thermometers, and it provides the basis for the definition of temperature.
First Law of Thermodynamics
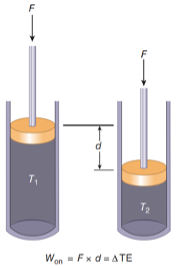
The first law of thermodynamics states that the total energy of a system can be increased by doing work on and/or applying heat to that system.
\[W_{\text{on}} + Q_{\text{to}} = \Delta E\]Examples:
- Pushing a shopping cart
- Work done by me is on the shopping cart
- Heating water
- Heat applied from the flame is to the water
References:
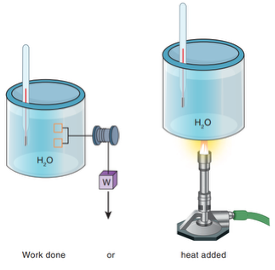
- Figure 5.2: Adding Work and Heat
- Figure 5.3: Using work to increase thermal energy
Demo: Adiabatic Compression - using work to increase thermal energy
Temperature
Temperature is a measure of the motion (average kinetic energy) of molecules.
- Hot temperatures → molecules move quickly
- Cold temperatures → molecules move slowly
Basis of Temperature Scales
- Fahrenheit scale: Based on human body temperature and the freezing point of a stable saline–alcohol solution
- Celsius scale: Based on the freezing and boiling points of water
| Reference Point | Fahrenheit | Celsius | Kelvin |
|---|---|---|---|
| Boiling point of water | 212°F | 100°C | 373 K |
| Human body temperature | ~100°F | 37.8°C | 310.8 K |
| Room temperature | 72°F | ~22°C | 295 K |
| Average global temperature | 59°F | 15°C | 288 K |
| Freezing point of water | 32°F | 0°C | 273 K |
| Freezing point of an alcohol–saline solution | 0°F | -17.8°C | 255.2 K |
| Absolute zero | -460°F | -273°C | 0 K |
Provide this table with just the basis temperatures. Fill in values as we discuss them. Have students convert the numbers in italics.
Converting Celsius to Fahrenheit: \(T^{^\circ\text{F}} = \frac{9}{5}T^{^\circ\text{C}} + 32\)
Class Activity 7 Part 1: In the temperature table, convert the temperatures given in Celsius to Fahrenheit.
Converting Fahrenheit to Celsius: \(T^{^\circ\text{C}} = \frac{5}{9}(T^{^\circ\text{F}} - 32)\)
Class Activity 7 Part 2: In the temperature table, convert the temperatures given in Fahrenheit to Celsius.
Absolute Zero
- If temperature is a measure of kinetic energy, could we decrease temperature until KE = 0?
- This theoretical temperature is called absolute zero
- It represents the temperature at which all molecular motion stops
Kelvin conversion: \(T^K = T^{^\circ\text{C}} + 273\)
Class Activity 7 Part 3: Complete the rest of the temperature table
States of Matter
- Gas: Molecules move around freely
- Liquid: Molecules move freely but are attracted to one another and stay close together
- Solid: Molecules are locked into a pattern; they still vibrate but remain in fixed positions