PHYS 1020 - Physics of Energy
Heat
Heat: Transfer of energy due to a difference in temperature
Types of heat — what is the result of the energy being transferred?
- Specific heat: Transfer of energy resulting in a change in temperature
- Latent heat: Transfer of energy resulting in a change in the state of matter
Reading:
- 5C-5F
Materials:
- Convection cycle tube, food coloring, food warmer/candle, matches
Activities:
Specific Heat
- Depends on mass — more mass means more energy is needed to heat it up
- Depends on the material — some materials require more energy to heat up
- Specific heats of common substances (Table 4.2)
How much heat is required to raise the temperature of 7 gallons of water from room temperature (70°F) to the boiling point (212°F)?
\[m = 7 \text{ gal} \times 8.34 \frac{\text{lb}}{\text{gal}} = 58.38 \text{ lb}\] \[Q = mc(T_f - T_0) = 58.38 \text{ lb} \times 1 \frac{\text{Btu}}{\text{lb °F}} \times (212 - 70)^\circ F\] \[Q = 8{,}389.96 \text{ Btu} \approx 8{,}745{,}908 \text{ J}\]
- 1 gallon of water weighs 8.34 lb
How much heat must 25 kg of water lose to cool from 16°C to 6°C?
\[\Delta T = -10°C\] \[Q = mc(T_f - T_0) = 25 \text{ kg} \times 4186 \frac{\text{J}}{\text{kg °C}} \times (-10°C)\] \[Q = -1{,}046{,}500 \text{ J}\]
If you apply 15,000 J of heat to 8.0 kg of copper $(c_{\text{Cu}} = 387 \text{ J/kg°C})$, how much does the temperature increase?
\[\Delta T = \frac{Q}{mc} = \frac{15{,}000}{8.0 \times 387} = 4.8°C\]
Latent Heat
- Sometimes heat is applied to change the state of matter
- When this happens, temperature does not change
- Depends on mass — more mass requires more energy
- Heat of vaporization: Energy absorbed to evaporate $(L_V)$
- Condensation releases energy $(-L_V)$
- $L_V = 2260 \text{ kJ/kg}$ for water
- Heat of fusion: Energy released to freeze $(-L_F)$
- Melting absorbs energy $(L_F)$
- $L_F = 335 \text{ kJ/kg}$ for water
How much heat is required to melt 5 kg of ice at 0°C?
\[Q = 5 \text{ kg} \times 335 \text{ kJ/kg} = 1675 \text{ kJ} = 1{,}675{,}000 \text{ J}\]
How much heat is required to evaporate 5 kg of water at 100°C?
\[Q = 5 \text{ kg} \times 2260 \text{ kJ/kg} = 11{,}300 \text{ kJ} = 11{,}300{,}000 \text{ J}\]
Heat vs. Temperature and State of Matter
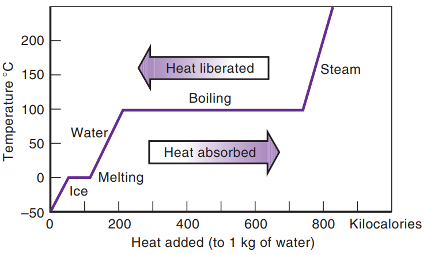
Figure 4.6 — Heat vs. Temperature and State of Matter for Water
As heat is absorbed/released
- temperature increases/decreases due to specific heat, or
- the substance changes to a higher/lower state of matter due to latent heat.
Why do you feel cold when you get out of a pool?
- Water on your skin begins to evaporate
- Evaporation requires energy (latent heat of vaporization)
- That energy comes from your skin
- As your skin loses energy, your temperature drops and you feel cold
How much heat is required to melt 5 kg of ice at 0°C, raise the temperature to 100°C, and evaporate it?
\[Q_1 = mL_F = 5 \times 335 \text{ kJ/kg} = 1{,}675{,}000 \text{ J}\]
- Melt the ice
- Raise the temperature
- Evaporate the water
Add them together to get the total
- Melt the ice
\[Q_2 = mc(T_f - T_i) = 5 \times 4186 \times (100 - 0) = 2{,}093{,}000 \text{ J}\]
- Raise the temperature
\[Q_3 = mL_V = 5 \times 2260 \text{ kJ/kg} = 11{,}300{,}000 \text{ J}\]
- Evaporate the water
\[Q = Q_1 + Q_2 + Q_3 = 15{,}068{,}000 \text{ J}\]
- Total energy
Question: Which stage takes the most energy?
Forms of Heat
How does heat occur? (That is, how is energy transferred?)
Radiation is the transfer of energy moves through electromagnetic waves
- Waves of electric and magnetic fields
- Associated with photons
- Examples: radio waves, microwaves, light, ultraviolet radiation, X‑rays
Convection is the transfer of energy via the motion of a fluid
- Example: Pot of soup — bottom heats up, warm fluid rises
Conduction is the transfer of energy from molecule to molecule
- Example: Touching a hot object
Conduction
Conduction is the transfer of energy from molecule to molecule. Think of this like billiard balls hitting each other.
Some materials conduct heat faster than others. Factors that come into the rate of heat:
- The thermal conductivity ($k$) is a value that indicates how quickly energy conducts through that material
- A larger $k$ means a lot of energy is transfered
- A larger area ($A$) allows more energy to flow than a smaller area
- A larger difference in temperatures ($T_f - T_0$) allows more heat to flow
- A larger thickness ($x$) prevents heat from flowing as quickly
Putting these together, we get the full Heat flow equation.
\[\frac{Q}{t} = \frac{kA(T_f - T_i)}{x}\]We often can’t control the temperature or the area. But we can control both the thickness and the conductivity by choosing different substances. The ratio between the thickness and the thermal conductivity is known as the R-Value.
\[R = \frac{x}{k} \qquad \frac{Q}{t} = \frac{A}{R}(T_f - T_0)\]- High (R) values → lower heat flow
- Low (R) values → higher heat flow
Ways to increase $R$:
- Increase thickness ($x$)
- Decrease conductivity ($k$)
Material comparisons:
- Metals and glass → high conductivity
- Rock and wood → lower conductivity
- Air and vacuum → extremely low conductivity
This is why fiberglass, double‑pane windows, and wool sweaters are good insulators—they trap air.
Fiberglass insulation has $k = 0.269 \tfrac{\text{ Btu}}{hr\cdot ft^2 \cdot \text{°F}}$. What is the R‑value if it is 3.5 inches thick?
\[R = \frac{x}{k} = \frac{3.5}{0.269} = 13.01\]
Reference:
Fiberglass Insulation – engineeringtoolbox.com
Units:
- If $x$ is in meters, $k$ must be in $\tfrac{W}{m\cdot K}$ → RSI value
- If $x$ is in inches, $k$ must be in $\tfrac{Btu\cdot in}{hr ft^2 °F}$
Class Activity 8: Passive Heating - Part 1: Conduction
Convection
Convection is the transfer of energy as a fluid flows, carrying the energy with it.
- Example: Pot of soup — warm water rises
Demo: Convection cycle
- We’ll focus on convection cycles in buildings to improve energy efficiency in a future lecture
Class Activity 8: Passive Heating - Part 2: Convection
Radiation
Radiation is the transfer of energy through electromagnetic waves.
- A wave is a disturbance that propagates through space
- Wave characteristics:
- Wavelength (determines type of wave)
- Frequency
- Velocity
Electromagnetic spectrum:
- Radio → Microwave → Infrared → Visible (Red–Violet) → UV → X‑rays → Gamma
Wave Equation
\[v = \lambda f\]Speed of light
\[v = 3.00 \times 10^8 \text{ m/s}\]Blue light has a wavelength of $\lambda = 500 \text{ nm}$. What is its frequency?
\[f = \frac{v}{\lambda} = \frac{3*10^8 m/s}{500 nm} = \frac{300,000,000 m/s}{0.000000500 m} = 6 * 10^{14} \text{ Hz}\]
Thermal Radiation
All objects emit radiation
- The type of radiation depends on temperature
Interactive simulation: Blackbody Spectrum (PhET)
Human body temperature ($~100^\circ F \approx 310 K$) indicates that we emit infrared radiation. This is why infrared sensors work.
Examples:
- Earth: ~20°C → infrared radiation
- Sun: ~6000°C → visible (green) light
Conduction, convection, and radiation all occur simultaneously and together transfer energy.
Class Activity 8: Passive Heating - Part 3: Radiation